Select the format in which animated models are to be shown:
- JSmol requires HTML 5.0, and can be slow
- Jmol requires Java to be installed on the client machine, but is sometimes much faster
Format currently selected: HTML5.0_JSmol
Mechanisms in Inorganic Chemistry
Introduction
- These pages deal with some substitution mechanisms for
transition metal complexes. Each is accessed via one of
the four links below, and should appear in a new window.
Each concerns a mechanism which goes via an intermediate
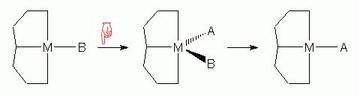
- Each page contains a reaction scheme with reaction arrows
leading to or from the intermediate. Clicking on a reaction
arrow produces a JSmol (or Jmol) model positioned beneath the
reaction scheme
- Hand symbols superimposed on the reaction scheme show which
models are switched on currently
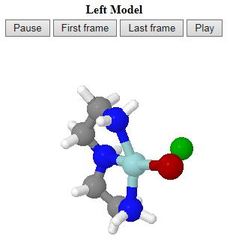
- One or two models side by side are allowed:
the left model is for a reaction leading from the starting
material to the intermediate,
while the right model is for the step going from the intermediate
to the product
- Clicking on the same arrow again, switches that model off
- Clicking the provided Animate button switches the program
to animate mode: starting with the leftmost model, a series of
animation frames are shown, representing the course of the
reaction. If two models have been selected, then when the
left model ends at the intermediate, the right model starts
automatically, carrying on from the intermediate to the product
- The models may be rotated by dragging over them using the
left mouse button.
Shift key and drag left mouse button upwards zooms out.
Shift key and drag left mouse button horizontally rotates about the screen Z
axis
- When two models are displayed, mouse control is for each one
separately
- Clicking the Animate button sets both models back to their default orientations and zoom
levels, and to the first frames of the animations.
To set up alternative viewpoints or zoom levels for both animations,
proceed as follows
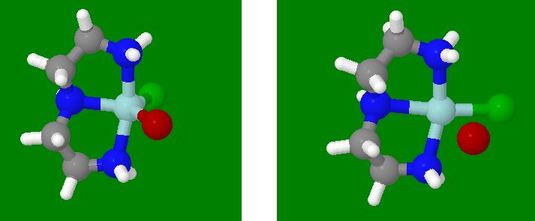
- Have both models selected
- Click the Animate button, then immediately click the Pause
button on the individual button bar for the Left Model, then click
its First frame button
- Adjust the viewpoint or zoom level for each model separately,
so that they match, as desired
- On the button bar for the Left Model, click Play
- Providing the program is in Animate mode, the Right Model
animation will start when the Left Model finishes, but now with
the desired orientation
- If the program is not in Animate mode, but is in its initial
First Frame Only mode, then the two models may still be animated
using their individual button bars, but there is no automatic
follow-on. This may be a desirable mode of use for
exploring details of the animations
Dissociative Substitution Mechanism for Octahedral Complexes
Animation of a Sn1 octahedral substitution
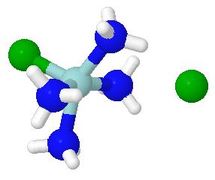
- Here, departure of the leaving group gives a square pyramidal
intermediate with the remaining substituents monodentate
- For the recombination step, the user may choose either
a simple nucleophilic attack on the metal, or one with
preliminary hydrogen bonding to ammonia ligands. This
may not be 'realistic', but it is interesting to watch
Enantiomers in an animation of a bis-chelate octahedral Sn1 substitution
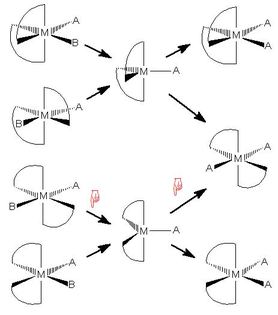
- Ethylenediamine ligands favour a 90° angle at the
metal, stabilising a trigonal bipyramidal 5‑coordinate intermediate rather than
a square pyramidal one, since the axial‑equatorial
angle in a trigonal bipyramid is 90°
- Only two trigonal bipyramidal molecules
[M(en)2A] are possible,
constituting a pair of enantiomers
- The clickable reaction scheme is in enantiomeric upper and
lower halves, connected only by trans‑[M(en)2A2],
which has a centre of symmetry so is not chiral.
All of the other species in the chart are chiral
- Any racemisation reactions must go via
trans‑[M(en)2A2] as an intermediate
- Two models may be selected only if they are in the same half
of the scheme
Associative Substitution Mechanism for Square Planar Complexes
- Associative substitution in a square planar complex goes
via a trigonal bipyramidal intermediate in which the entering
group is equatorial
- In contrast to main groups systems, in which attacking and
leaving groups are in axial positions in 5‑coordinate intermediates,
it is one of the equatorial groups which leaves in substitution
reactions in transition metal complexes
Animation of Replacement of a Unique Ligand, with the Remaining Positions
Occupied by Diethylenetriamine, a Tridentate Ligand
- Diethylenetriamine gives two adjacent 90° angles at the
metal in its complexes, making it ideal for the square planar
geometry
- In a trigonal bipyramidal intermediate, diethylenetriamine
must bridge the two axial positions via one equatorial position
- This leaves the remaining two equatorial positions for the
attacking and leaving groups, in agreement with the principle
outlined above
Animation of Replacement of a Ligand, with the Remainder Monodentate
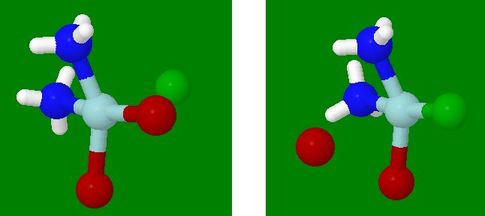
- Without the tridentate ligand to control the positioning
around the trigonal bipyramidal intermediate, the principle,
above, of entering and leaving at equatorial positions still holds
- The net result is that the entering group replaces the leaving
group in the square planar complex, leading to retention of
configuration
- In the second and third examples shown in the clickable
reaction scheme,
cis‑ML2 remains cis,
and trans‑ML2 remains trans
- If two models are selected, they must be for the same
reaction sequence
Select background colour for animations:
- Black may be best for use with a projector
- White is better for screenshots for possible printing
Colour selected = White
About Mechanisms in Inorganic Chemistry





