Pretargeted imaging and therapy
Development of new pretargeting strategies
Antibodies labelled with radioisotopes are frequently used to detect and characterise tumours in vivo in a non-invasive manner by PET imaging. However, antibodies suffer from prolonged residence times within the circulation and consequently high tumour-to-blood contrast ratios are only observed 5-7 days after their administration. Furthermore, the patient is also exposed to an increasing radiation dose over this timeframe. To address these problems, an imaging strategy known as ‘pretargeting’ has been developed.
Pretargeting involves the sequential administration of (i) an antibody ‘primary agent’ and (ii) a low molecular weight radiolabelled ‘secondary agent’ which is capable of binding to the primary agent at the tumour surface. In practical terms, the antibody – which has inherently slow pharmacokinetics – is first permitted sufficient time to reach an optimal tumour-to-blood ratio (days). At this point, the radioactive secondary agent – which has fast pharmacokinetics – is administered and undergoes a rapid and selective ‘click’ reaction with the antibody at the tumour site (minutes). This results in an improvement of imaging contrast at earlier time points after administration of the radioactive agent and leads to a significantly reduced radiation dose to the patient.
During my previous appointment at Oxford University with Prof. Bart Cornelissen, we succeeded in developing a novel enzymatic pretargeting strategy which has demonstrated striking efficacy in models of colorectal cancer. Now, the JCK group is advancing this technology in a new direction, using bioorthogonal chemical reactions which take place inside a cancer patient to activate cancer-killing warheads localised at the tumour surface.
You can read more about this in our recent papers on this topic, here, here, and here!
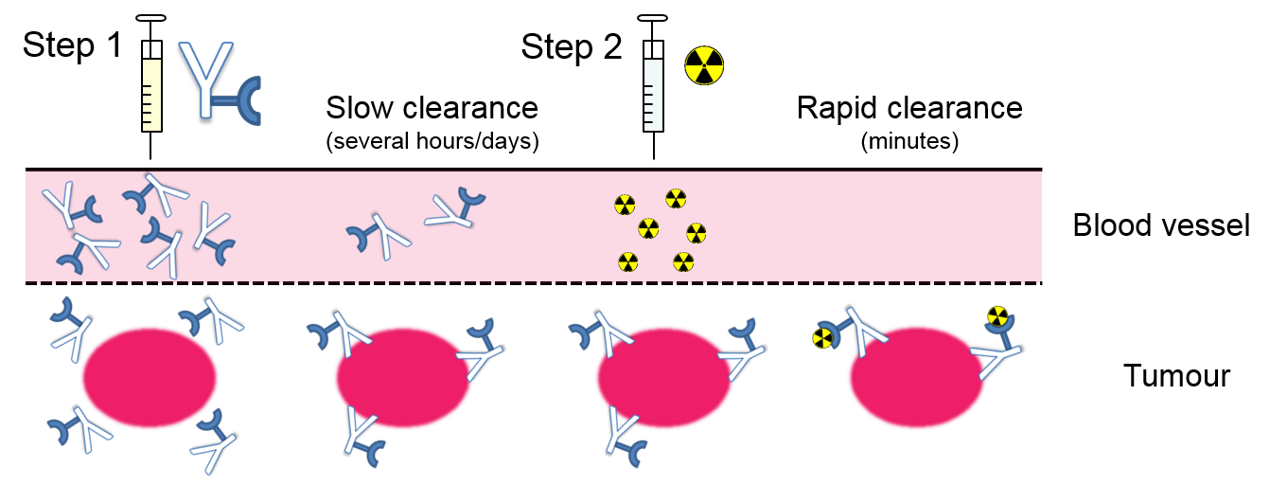 Pretargeted imaging is a 2-step strategy which results in high quality images soon after administration of the radiotracer and reduces the radiation dose to the patient
Pretargeted imaging is a 2-step strategy which results in high quality images soon after administration of the radiotracer and reduces the radiation dose to the patient